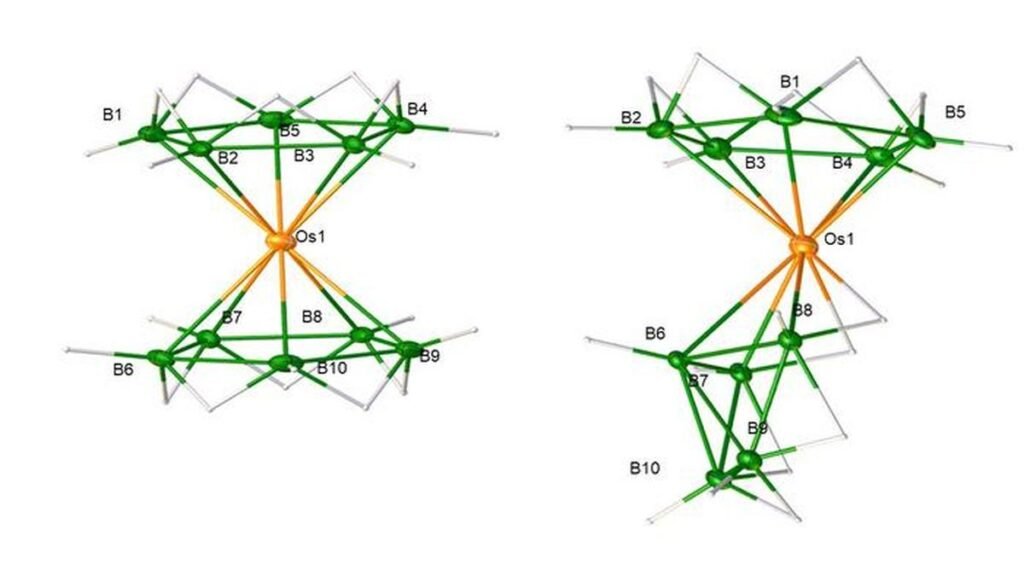
An illustration of the new boron-osmium sandwich and its alternative configuration (isomer).
| Photo Credit: Suvam Saha
A compound called ferrocene has been a landmark of chemistry for over seven decades. It consists of an iron atom sandwiched between two flat carbon rings. Its discovery launched the field of organometallic chemistry, with numerous applications in materials science and medicine. Since then, chemists have been trying to create a version of this sandwich that contains no carbon atoms.
In a new study in Science, researchers from IIT-Madras and the Indian Institute of Science (IISc), Bengaluru, announced they had finally achieved this goal: a stable, carbon-free analogue of ferrocene using boron rings and osmium.
“Just as ferrocene started a new era in organometallics, these results will start a new era in inorganometallics and will soon be a part of textbooks of inorganic chemistry,” study co-author and IIT-Madras professor Sundargopal Ghosh said in a release. “Our efforts are on to study the reactions of these new compounds.”
Boron is next to carbon on the periodic table and can form similar ring structures. While scientists had previously created sandwiches containing both carbon and boron, a purely inorganic version has been elusive.
The team turned to computer modelling to predict which metal would best stabilise a boron sandwich, and zeroed in on osmium. To create the complex, the team reacted a polymeric osmium-bromine precursor compound with an excess of a borane-dimethyl sulphide reagent. Then they heated the mixture to 100 °C for eight hours and isolated the product as a colourless solid. Finally, they used X-ray diffraction and nuclear magnetic resonance spectroscopy to check the atoms’ arrangement.
The X-ray analysis revealed a perfect sandwich, with a single osmium atom between two parallel, five-membered boron rings. But unlike the flat carbon rings in ferrocene, the boron rings used bridging hydrogen atoms between the boron atoms. These bridges redirected the ring’s electron orbitals towards the metal, creating a bond even stronger than the one in ferrocene. This could pave the way for new catalysts that are stable at much higher temperatures, leading to the more efficient production of pharmaceuticals.
According to the paper, the discovery establishes that the iconic sandwich architecture is not exclusive to carbon-based organic chemistry. “With the renaissance in the 2D chemistry of boron during the last decade — with borophenes, bilayer borophenes, and multilayer borophenes on the horizon — the possibility of metal sandwiched/intercalated bilayers and multilayers will be a reality soon,” Eluvathingal D. Jemmis, IISc chemistry professor and one of the corresponding authors, said.
Published – May 13, 2026 08:00 am IST